Instructions for Disposable Tissue Closure Clip
1.Tissue closure clip name, model, specification
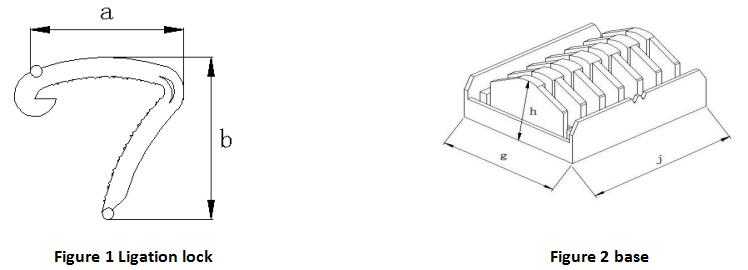
Table 1 Basic Dimensions of Closure Clamps Unit is mm
| Size
model |
a |
Tolerance | b | Tolerance | g | Tolerance | j | Tolerance | h | Tolerance |
| P-ZJ-S | 9.5 | ±1 | 9.4 | ±1 | 26.8 | ±1.5 | 32.5 | ±2 | 14.4 | ±1 |
| P-ZJ-M | 11 | 13 | 26.9 | 32.6 | 16.9 | |||||
| P-ZJ-L | 15.5 | 16.5 | 28.7 | 34.1 | 17 |
2. Tissue Closure Clip Performance
Ligation range of ligation lock: diameter of ligation tissue: large size: 6mm~10mm; medium size: 3mm~6mm; In the closed state, it should be able to withstand a tension of 5N without deformation, loosening or breaking, and the ligature lock must not fall off after being installed in the base.
3. The main structural components of the tissue closure clip
The disposable tissue closure clip consists of a ligation lock, a fixed cover and a base.
The material of the ligation lock is POM, and the material of the fixed cover and base is ABS.
4. Scope of application of tissue closure clip
It is suitable for clamping tissue bundles in clinical abdominal surgery (removed after surgery)
- The structure of the tissue closure clip
6. tissue closure clip contraindications
none
7. Tissue closure clip installation
none
8. Instructions for use of tissue closure clips
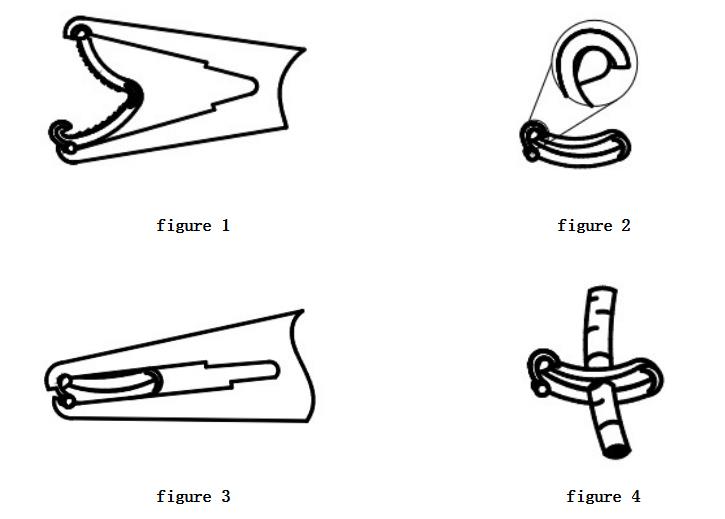
Disposable tissue closure clips are suitable for the sizes of blood vessels and tissue structures shown in the attached drawings, and clinicians should judge and select appropriate disposable tissue closure clips.
①. Place disposable tissue closure clips:
(1) Place the tissue forceps, grasp the tissue forceps tightly, and place the head of the tissue forceps accurately into the base of the tissue closure clip, confirm that the head is perpendicular to the base, and gently press the tissue forceps until a click is heard , do not force the tissue forceps Anjou base or the upper part of the tissue closure clip, the tissue closure clip can enter and exit the base easily;
(2) Remove the tissue forceps from the base to ensure that the tissue closure clips are safely clamped in the occlusal surface of the tissue forceps (as shown in Figure 1). At this time, the base must be held to ensure that the tissue closure clips can be smoothly removed from the base;
(3) During use, as shown in the figure (Figure 1), position the single tooth of the tissue closure clip. At this time, the operator can confirm the ligated structure with the naked eye;
(4) Position the tissue closure clip around the ligated blood vessel and body cavity tissue (as shown in Figure 2), and apply sufficient force on the handle of the tissue forceps so that its occlusal surface is closed and the tissue closure clip is locked (as shown in Figure 2). 3), loosen the handle of the tissue forceps, and let the tissue forceps return to a sufficiently large position.
(5) Note: If the blood vessels and body cavity tissue are stripped (as shown in Figure 4), leave the end edge about 2-3 mm away from the tissue closure clip, and do not use the end of the tissue closure clip as a guide for incision.
(6) After the operation, remove the ligation lock of the tissue closure clip.
9.Tissue closure clip maintenance and maintenance methods
The packaged product should be stored in a well-ventilated room with a relative humidity not exceeding 80%, no corrosive gas, ventilation and light.
10.Tissue closure clip Expiration date
It is valid for three years after being sterilized by ethylene oxide, and the expiry date is shown on the label.
11.Tissue closure Parts list
none
12.Tissue closure Precautions and warnings
(1). When using this product, aseptic operation specifications should be strictly implemented
(2).Please check the packaging of this product carefully before use, and stop using it if it is damaged;
(3). This product is sterilized by ethylene oxide, and it is provided for clinical use in the form of a sterile product.
(4). This product is for one-time use and cannot be sterilized repeatedly;
(5). Please check whether the product is within the validity period before use. After being sterilized by ethylene oxide, the validity period is three years. Products beyond the validity period are strictly prohibited.
(6). This product is intended for use by physicians who have received professional knowledge and technical training on staplers or under the guidance of physicians with relevant experience;
(7). After use, it shall be destroyed according to the requirements of environmental protection or hospitals, and shall not be discarded at will.
(8). See label for production date
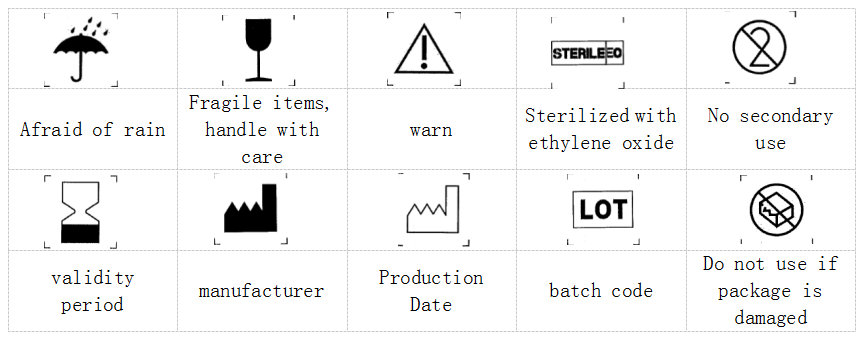
(9). Explanation of graphics, symbols and abbreviations used in packaging and labels.
Related Products
Post time: Feb-22-2023